
Using the Mathcad routine developed in Example 1.9 gives. The molar volume of ethanol is calculated from equation (1-48) VbB 60.9 cm3/mol. Which, when simplified, gives us an answer of 331 g per mole or the molar mass. According to restriction 3 mentioned above, the molar volume of the acetic acid to be used in equation (1-54) should be VhA - 2 x 62.4 124.8 cm3/mol. We get that molar mass is equal to three g over the 9.72 times 10 to the negative third moles. Rearranging this equation to isolate the three g for mila moles, we get nine point we get 9.72 times 10 to the negative third moles Is equal to that three g per molar mass, isolating molar mass. We're gonna take this and we're going to divide that by 0. That's how we're gonna solve for a number of moles. That we have, we're gonna place this over the molar mass and we're going to see wine a bit. And we're going to say that that is molds, pour kilogram and then we're gonna equal that to our three g of D. And so what we're gonna do is we're gonna take that 0.4536. When we saw for em we're going to get the m equals 0.4536 morality morality is simply moles per kilogram. 23☌ and the constant or the freezing point malakal constant for Benzene is 5.07 degrees of Celsius morality. We're then going to take our freezing point depression. or a mixture of 95. EtOH also called ethyl alcohol, grain alcohol. And what we're gonna do is we're going to take the number subjected by the 5.27 That we see in this question stem to give us 0.23☌. Molar mass: 46.069 g·mol 1 Appearance Colourless liquid Odor: Methanol-like. We do that by taking The freezing point of Benzene which is 5.5☌. So the first thing I wanna do is we want to calculate the freezing point depression. And we're supposed to calculate the molar mass of D. In 30.2 kg of benzene freezes at 5.27 degrees Celsius. It's a solution composed of three g of D. 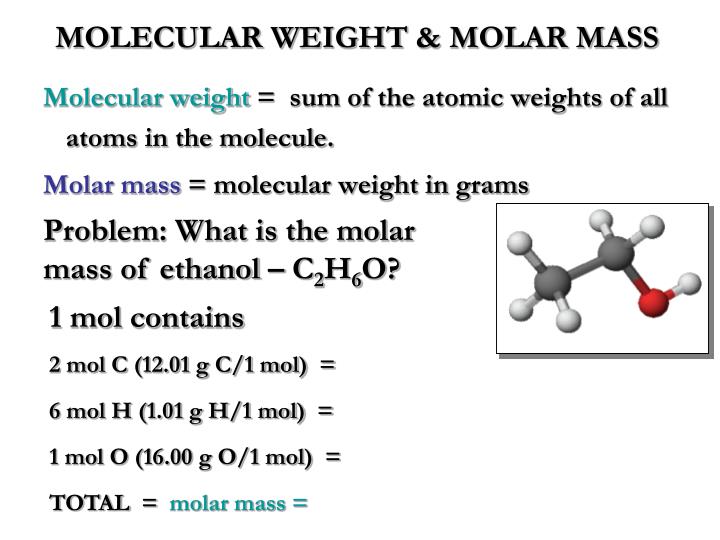
We are being asked to solve the following problem. In this work, we study the molar mass dependence of the thermodiffusion of polyethylene oxide at different temperatures in ethanol, water/ethanol mixture (c. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.Hello everyone today. These relative weights computed from the chemical equation are sometimes called equation weights. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. 
The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. This site explains how to find molar mass. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. Known Mass of ethanol 1.55 g Molar mass of ethanol 46.1 g/mol Mass of water 200 g c p water 4.18 J/g o C Temperature increase 55 o C Unknown Step 2: Solve. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.Ī common request on this site is to convert grams to moles. Step 1: List the known quantities and plan the problem. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.įinding molar mass starts with units of grams per mole (g/mol).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
